Research
Effects of Systemic Inflammation on Host Morbidity and Mortality

The Jung Kim Lab is seeking to answer the fundamental question in cancer biology; "How do tumors actually kill hosts?"
Tumors disrupt host physiology locally and systemically. Locally, tumors damage adjacent host tissues via direct contact. Systemically, tumors release many factors including cytokines, hormones, and metabolites, which travel through the circulation and affect organs at a distance. These systemic effects are also called paraneoplastic syndromes or paraneoplasias, and the mechanisms underlying here are still unclear.
We mainly focus on the systemic effects of tumors caused by tumor-secreted cytokines or hormones. Tumors can disrupt host organs at a distance via systemic effects, also called paraneoplastic syndromes or paraneoplasias. Using powerful Drosophila models, we found that tumor-secreted cytokine, fly IL-6, disrupts the blood-brain barrier (BBB) in the brain, leading to host death. Furthermore, these exciting phenotypes are recapitulated in mice (Developmental Cell, 2021). Strikingly, blocking systemic inflammation caused by fly IL-6 partially rescued BBB damage as well as reduced lifespan, in both flies and mice.


This is the beginning of understanding the molecular and cellular mechanisms of paraneoplastic syndromes. The goal of the Jung Kim Lab is to figure out what the tumor-secreted factors are and their functions and targets. To elucidate mechanisms here, we focus on the following research projects.
Project 1: Systemic effects of tumors on renal failure
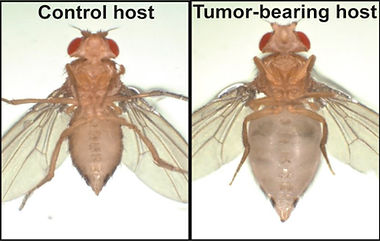
Tumor-bearing flies exhibit the bloating phenotype, which is excess fluid retention in the belly. We currently hypothesized that tumor-dependent systemic effects perturb the physiology of the renal tubule, functionally analogous to the kidney in mammals. Using powerful genetic experiments in vivo and ex vivo, we are investigating mechanisms in detail.
Project 2: Cellular and molecular mechanisms of blood-brain barrier damage by systemic inflammation
We are further investigating mechanisms by which Jak/STAT signaling pathway regulates the tight junctions in BBB. Through the RNAi-based genetic screen, we are studying what the target genes of STAT in the BBB are. Further, we investigate the cellular functions of putative candidates in terms of BBB opening.

Project 3: Novel tumor-secreted factors regulating host morbidity and mortality

Ovarian Carcinoma
Using a newly developed genetic ovarian carcinoma model, we are performing an RNAi-based screen to identify tumor-secreted factors modulating host morbidity and mortality.